RBGPF
0.5000

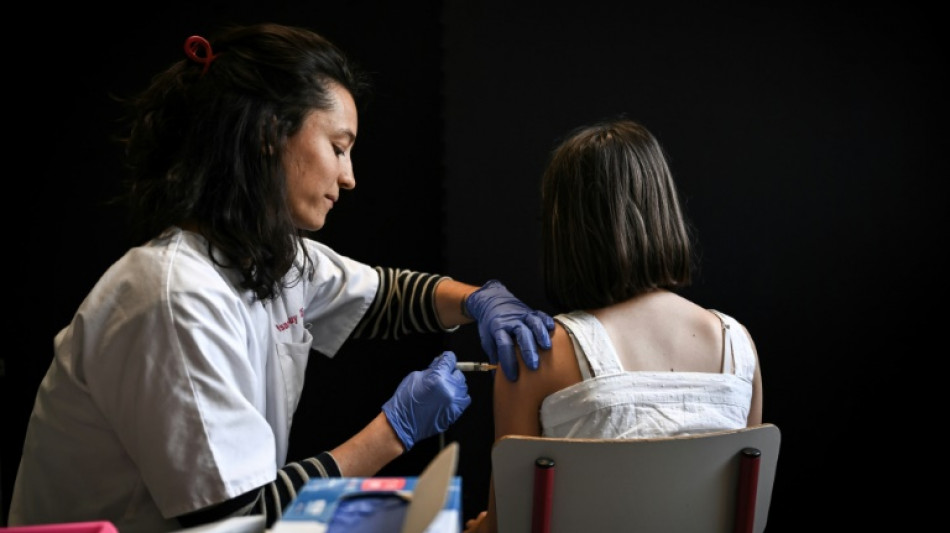
A new study published Thursday by researchers at the US Centers for Disease Control and Prevention (CDC) highlights the powerful impact of the human papillomavirus (HPV) vaccine, first approved in 2006, in preventing precancerous lesions detected through cervical screening.
HPV is the most common sexually transmitted infection, and certain strains can lead to cervical, vulvar, vaginal, and other cancers later in life. Each year, around 14 million Americans contract HPV, and the virus is responsible for approximately 10,800 cases of cervical cancer annually.
Since 2008, the CDC's Human Papillomavirus Vaccine Impact Monitoring Project has tracked trends in precancerous cervical lesions to evaluate the vaccine's effectiveness.
The findings are striking. Among women aged 20–24 screened for cervical cancer between 2008 and 2022, rates of moderate-to-high-risk precancerous lesions dropped by about 80 percent.
For women aged 25–29, the decline was 37 percent -- a smaller reduction the researchers attributed to many in this age group receiving the vaccine later in life as part of a "catch-up" vaccination effort, which is less effective.
In the US, the HPV vaccine was first approved in 2006 for girls and women, with boys and men added to the recommendations in 2011. Today, the CDC recommends routine HPV vaccination for boys and girls ages 11–12 to prevent infections that can lead to cancer later in life.
HPV includes over 200 different strains, some of which cause genital warts. Two particular strains -- 16 and 18 -- are classified as high-risk for cancer.
While cervical cancer is treatable if caught early, it is still responsible for around 4,400 US deaths per year.
The US Preventive Services Task Force recommends women ages 21 to 65 receive a Pap smear test once every three years and that women over 30 get high-risk HPV test every five years.
"The data are consistent with a considerable impact from the US HPV vaccination program on cervical precancers, with the largest decreases in the youngest age group for which benefit of vaccination would first be observed," wrote the authors in the CDC's Morbidity and Mortality Weekly Report.
They added that as vaccinated women age, further declines in cervical precancers are expected in older groups.
One limitation of the study was that individual vaccination status was not recorded, meaning the study could not directly confirm causality. However, the authors noted that "no other plausible explanations" have been identified for the sharp decline in precancers.
The study's positive findings come as outspoken anti-vaccine advocate Robert F. Kennedy Jr. begins his tenure as US health secretary.
Under his leadership, the Food and Drug Administration abruptly canceled a scheduled meeting with experts on Wednesday to discuss the development of next year's flu vaccines.
H.El-Hassany--DT